- Blog
- Nhl 20 controls
- Welcome to hanwell game play in hours
- Atom definition chemistry
- Illuminate meaning
- Ringo starr now
- Diy double sided magnets
- Girulian denjin makai 2
- Haddock fish and chips near me
- Onedrive for mac not syncing all files with office 365
- Scorch star wars
- Bartleby the scrivener pdf
- Johannes brahms compositions

This means a neutral oxygen atom has eight protons and eight electrons. An oxygen atom (O) has an atomic number of eight (8). If a hydrogen atom is neutral, it must also have one electron. This means a hydrogen atom has one proton. For example, hydrogen (H) has an atomic number of one (1). On the periodic table, the atomic number is usually given as the whole number above the symbol for the element (see Fig. The atomic number is equal to the number of protons in an element. When the number of protons and electrons in an atom are same, the charges cancel out, or counteract each other.Įvery atom of a particular element has the same number of protons. An uncharged atom is electrically neutral because electrons and protons have opposite charges of equal sizes. A single uncharged atom has an equal number of protons (+) and electrons (–). Some atoms exist naturally as neutral, or uncharged, atoms. The subatomic particles in an atom determine the properties of the atom. This means that it would take one hundred trillion (100,000,000,000,000) electrons put side-by-side to equal a centimeter! Electrons are about one-thousandth (1/1000) the diameter of a proton or neutron. This means it would take about one hundred billion (100,000,000,000) protons or neutrons put side-by-side to equal a centimeter. Protons and neutrons are both about one-thousandth (1/1000) the diameter of a hydrogen atom. One hundred million (100,000,000) hydrogen atoms put side-by-side equals about a centimeter. Chemical properties include the specific ways that atoms and molecules react and the energy that they release or use in these reactions. The number of electrons and protons in an atom determines its chemical properties. This force got its name because it is strong enough to overcome the force of the positively charged protons repelling each other. The strong force holds neutrons and protons together in the nucleus. The attraction between negatively charged electrons and positively charged protons in an atom give the atom its structure. Electrostatic forces also hold electrons and protons together in the atom. 2.11).Įlectrostatic forces hold atoms together in molecules-like the two hydrogen atoms held together in H2 gas. Electrons exist in regions called shells outside of the atom’s nucleus (Fig. Protons and neutrons occupy the nucleus, or center, of the atom.

For example, water (H 2O) has three atoms, two hydrogen (H) atoms and one oxygen (O) atom. Molecules of compounds have atoms of two or more different elements. All elemental molecules are made of atoms of a single element.

Another form of oxygen, ozone (O 3), has three atoms, and sulfur (S 8) has eight atoms. Hydrogen (H 2), oxygen (O 2), and chlorine (Cl 2) molecules, for example, each contains two atoms. Other elements contain two or more atoms in their molecular form (Fig. 2.8) is an example of a monatomic element. Some elements are monatomic, meaning they are made of a single ( mon-) atom ( -atomic) in their molecular form. A hundred million (100,000,000) hydrogen atoms put side-by-side is only as long as one centimeter!
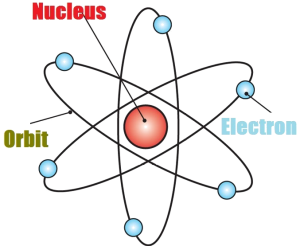
The simplest structural unit of an element is an atom. The properties of elements and compounds are determined by their structures.
- Blog
- Nhl 20 controls
- Welcome to hanwell game play in hours
- Atom definition chemistry
- Illuminate meaning
- Ringo starr now
- Diy double sided magnets
- Girulian denjin makai 2
- Haddock fish and chips near me
- Onedrive for mac not syncing all files with office 365
- Scorch star wars
- Bartleby the scrivener pdf
- Johannes brahms compositions
